
Translation in eukaryotic systems occurs predominantly through 7-methylguanosine (m 7G) cap-dependent pathways 1. The assay can be widely applied to distinct translation systems and should broadly benefit in vitro studies of cap-dependent translation kinetics and translational control mechanisms.
This assay enables tracking of individual cap-dependent translation events and resolves key translation kinetics, such as initiation and elongation rates.
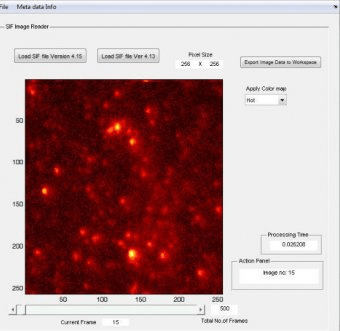
Here, we present a protocol for establishing this assay, including mRNA and PEGylated slide preparations, real-time imaging of translation, and analysis of single molecule trajectories. By imaging the binding and dissociation of antibodies to and from nascent peptide–ribosome–mRNA complexes, the translation progression on individual mRNAs can be tracked. The assay is based on fluorescently labeled antibody binding to nascent epitope-tagged polypeptide. Recently, we developed an in vitro assay to measure cap-dependent translation kinetics with single-molecule resolution. While various biochemical and genetic approaches have allowed extensive studies of cap-dependent translation and its regulation, high resolution kinetic characterization of this translation pathway is still lacking. Cap-dependent protein synthesis is the predominant translation pathway in eukaryotic cells.
